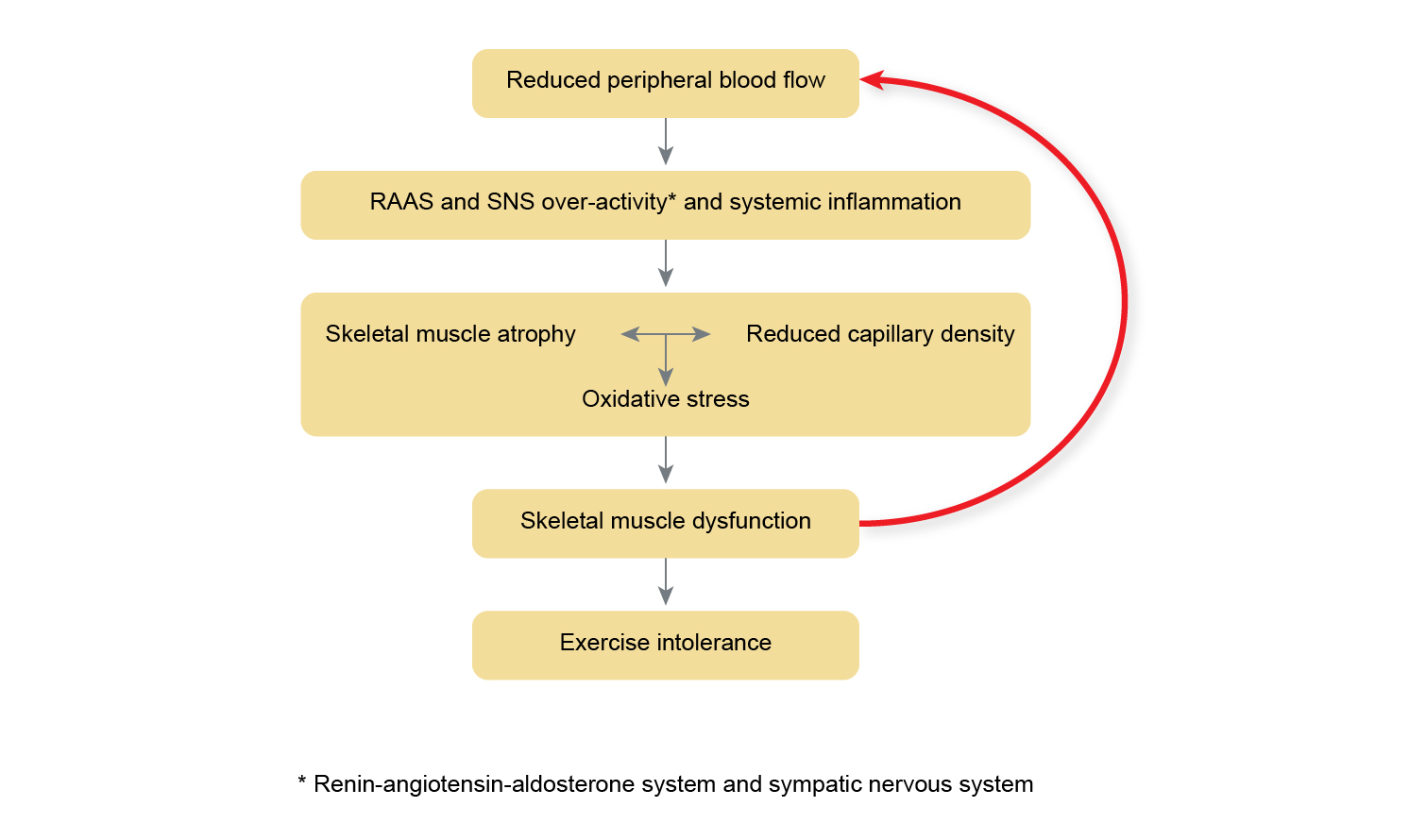
There is strong evidence supporting the benefits of exercise training in people with heart failure (HF) and all patients should be referred to a specifically designed exercise program, if available.
Benefits of exercise training include improved quality of life, cardiorespiratory fitness and reduced symptoms of fatigue and dyspnoea. The gold standard measure of aerobic fitness, peak oxygen consumption (VO2 peak), is strongly associated with survival in HF patients and typically increases by 15-20% over a training period of 8-12 weeks, with some studies showing increases of up to 30%.
These improvements in cardiovascular fitness are predominantly due to peripheral adaptations, such as improved oxidative capacity of skeletal muscle and enhanced peripheral blood flow following aerobic training, and muscle hypertrophy following resistance training.
Resistance training complements aerobic exercise, resulting in strength increases of up to 40%, depending on the characteristics of the training intervention. In a systematic review and meta-analysis published in 2022, resistance training was found to elicit significant improvements in VO2 peak and 6MWD, when compared to controls. [#fisher-s-smart-na-pearson-mj ]
Recent work has raised the possibility that high-intensity interval training may offer superior benefits to HF patients compared with low-to-moderate intensity continuous aerobic training with regard to VO2 peak. However, results are not conclusive and should be interpreted with caution.
Psychological benefits of exercise training include reduced depression and anxiety, and improved quality of life. HF patients who exercise regularly also have better clinical outcomes. The largest exercise training study in HF, the HF-ACTION trial, randomised 2,331 patients to aerobic exercise or usual care. After adjusting for predetermined prognostic factors, exercise training was associated with a 15% reduction in all-cause mortality and decreased the number of hospitalisations due to HF.
Is exercise safe for patients with heart failure?
Until recently, research studies have been conducted predominantly in patients with stable symptoms (New York Heart Association [NYHA] Class I-III), HFrEF and young males, and in these cohorts, moderate-intensity exercise is well tolerated. In the many studies published to date, there have not been any reported deaths directly related to the effects of exercise training.
Far fewer studies have been published related to frail, older patients with heart failure, however available data suggests that these patients demonstrate high levels of adherence and report improvements in short performance physical battery, 6MWT, frailty scores and quality of life [#kitzman-dw-whellan-dj-duncan-p.-et-al]. Studies also suggest that those with more severe symptoms are most likely to demonstrate greater mortality and morbidity benefit from supervised exercise training compared to those with fewer symptoms and therefore should not be excluded from exercise training programs.
 Pathophysiology
Pathophysiology
 Treatment & Management
Treatment & Management
 Exercise
Exercise
 Medications
Medications
 Psychosocial Issues
Psychosocial Issues
 Patient Education
Patient Education
 Behaviour Change
Behaviour Change
 Clinical Indicators
Clinical Indicators
 Pathophysiology
Pathophysiology
 Treatment & Management
Treatment & Management
 Exercise
Exercise
 Medications
Medications
 Psychosocial Issues
Psychosocial Issues
 Patient Education
Patient Education
 Behaviour Change
Behaviour Change
 Clinical Indicators
Clinical Indicators
 Pathophysiology
Pathophysiology
 Treatment & Management
Treatment & Management
 Exercise
Exercise
 Medications
Medications
 Psychosocial Issues
Psychosocial Issues
 Patient Education
Patient Education
 Behaviour Change
Behaviour Change
 Clinical Indicators
Clinical Indicators